 
d S d Q T only applies to energy transfer via heat transfer, not work. For elements, the standard molar entropy is. 
That means that entropy of formation is pretty useless because you need to know the actual entropy of the standard state of components to know what the entropy of the species is. Gibbs, whose work was translated into French by Le Chatelier, lived with his sister and brother-in-law until his death in 1903, shortly before the inauguration of the Nobel Prizes. Since work involves both a change in volume and an energy transfer, the entropy change due to work should have two terms: (1) entropy change due to energy transferred via work (2) entropy change due to volume change. We dont use this quantity because entropy is not an energy, therefore, you cannot set the standard state entropy to 0. His series of papers entitled “On the Equilibrium of Heterogeneous Substances” was the foundation of the field of physical chemistry and is considered one of the great achievements of the 19th century. He was appointed professor of mathematical physics at Yale in 1871, the first such professorship in the United States. All spontaneous changes cause an increase in the entropy of the universe. reversible (system is at equilibrium) Definition: The Second Law of Thermodynamics. nonspontaneous (spontaneous in opposite direction) Suniv 0. For more information on the entropy formula and the effect of entropy on the spontaneity of a process. Problem: Calculate the entropy change of 1 mole of an ideal gas that undergoes an isothermal transformation. Table 7.3.1: The Second Law of Thermodynamics. As the gas expands in a system, entropy increases. 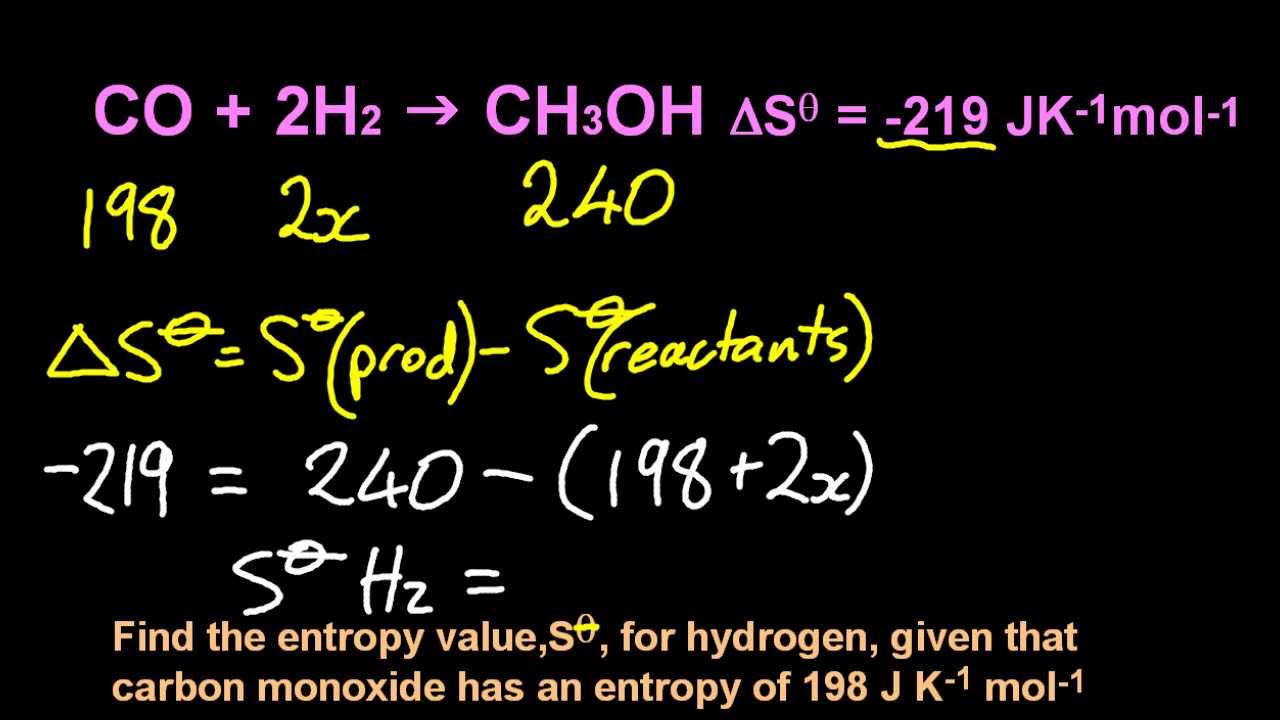
For any chemical reaction, the standard entropy change is the sum of the standard molar entropies of the products minus the sum of the standard molar entropies of the reactants. S (1/T) 12PdV (nR) 12(1/V)dV nRln(V2/V1). In 1863, Gibbs was awarded the first engineering doctorate granted in the United States. About Transcript The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. The temperature in this equation must be. \): Relation between Process Spontaneity and Signs of Thermodynamic Properties Δ S univ > 0īorn in Connecticut, Josiah Willard Gibbs attended Yale, as did his father, a professor of sacred literature at Yale, who was involved in the Amistad trial. The equation for the change in entropy, S, is. Using this equation it is possible to measure entropy changes using a calorimeter. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
