 I think that this orbital is responsible for the change in bond angle from the predicted 109.5. The HOMO is the interesting one, it is the 3pz orbital of P bonding with the all bonding combo of the 1s H orbitals. The others are two degenerate orbitals with one node (the 2 3p orbitals of P bonding with the antibonding and nonbonding combination of the 3 1s orbitals of H). The lowest valence orbital is all bonding with the 3s orbital of P and the 1s orbitals of H. The MOs look a great deal like ammonia's MOs except the 2s and 2p orbitals do not participate in bonding at all. Here are some results using RMP2/6-31g(d) I ran a calculation on PH3 using Gaussian, just to look at the MOs. In this case, they don't even spend most of their time on one atom because there is no electronegativity difference. 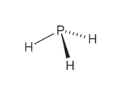 
Electrons delocalize throughout the molecule, they don't just stay in one place. There really is no such thing as lone pairs. I just have some problems with "lone pairs". If anyone has anything further to add, please do so. Induced dipole-dipole van der Waals interactions, that are present between all molecules (whether polar or non-polar), would play a significant role in the intermolecular attractive forces for PH3. Therefore, you would expect the permanent dipole-dipole interactions to be weak, unlike the stronger hydrogen bonding in NH3. Hence, due to the particular nature of the PH3 molecular orbitals and the trigonal pyramidal molecular geometry, specifically that the lone pair occupying the 3s orbital and thus being relatively closer to the P atom than the bond pairs between P and H, there is consequently a slightly greater electron density and a slightly partial negative charge on the P atom (even as P and H have the same electronegativity), and hence a slight dipole moment, making PH3 a slightly polar molecule. In conclusion, the lone pair is close to the P nucleus, and hence P has slightly increased electron density, which has an effect on polarity of the molecule.) spend more time nearer) to the nucleus, compared to the electrons in the p orbitals. Recall that the electrons in the s orbital, on average, are closer (ie. Notice then, that the s orbitals are largely not involved in bonding, ie. 
Since in PH3 the angles are much closer to 90 deg than 109.5 deg, you would expect the P-H bonds to be mainly sigma bonds from the overlap of p electrons (from P) and s electrons (from H). Whilst sp3 hybridized orbitals have 109.5 deg angles. Recall that p orbitals are px, py and pz, 90 deg angles. (edited : For those who might be confused, I will give a simplified explanation of the statement "the almost orthogonal bond angles lead to the conclusion that in PH3 the P-H bonds are almost entirely pσ(P) – sσ(H) and the lone pair contributes only a little to the molecular orbitals". 
This electronic structure leads to a lack of nucleophilicity and an inability to form hydrogen bonds. The high positive chemical shift of the P atom in31P NMR spectrum accords with the conclusion that the lone pair electrons occupy the 3s orbital and so are close to the P atom. The low dipole moment and almost orthogonal bond angles lead to the conclusion that in PH3 the P-H bonds are almost entirely pσ(P) – sσ(H) and the lone pair contributes only a little to the molecular orbitals. In contrast, the dipole moments of amines decrease with substitution, starting with ammonia, which has a dipole moment of 1.47 D. The dipole moment is 0.58 D, which increases with substitution of methyl groups in the series: CH3PH2, 1.10 D (CH3)2PH, 1.23 D (CH3)3P, 1.19 D. The length of the P-H bond 1.42 Å, the H-P-H bond angles are 93.5°. > PH3 is a trigonal pyramidal molecule with C3v molecular symmetry. However, note that because of the following (from Wikipedia) : If anyone has anything further to add, please do so.Īs the electron geometry is tetrahedral (the molecular geometry is trigonal pyramidal), you might expect the P bonding orbitals to be sp3 hybridized, in accordance to the bond angles. So after thinking about it a bit, and from the Wikipedia info, the following are my thoughts. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
